The broker representing the 60-foot vessel was dumbfounded. “How could that be?” he asked. “They are stainless steel, and stainless steel doesn’t rust.” I had just told him that the boat’s 4-year-old stainless-steel water tanks were corroding and leaking — they had to be replaced. It represented an invasive and costly undertaking, and one that could have easily been avoided.
In a sense, he was right. Stainless steel doesn’t rust, not in the commonly understood sense; it does, however, corrode. He could also be forgiven, to some degree, for his incredulity. Many marine industry professionals — and most boat owners — are under the mistaken impression that stainless steel is impervious to any type of decay or corrosion. In fact, nothing could be further from the truth. It’s an important issue for voyagers, who often are relying on stainless-steel parts when they are voyaging offshore.
Stainless steel(s)
Since its accidental discovery in 1913, stainless steel has become extremely popular in fields beyond the marine and boatbuilding trades. Everything from automobiles and jet turbines to skyscrapers (incidentally, a “skyscraper” was a sail before it was a building) and flatware utilize massive quantities of this metal that seemed, at the time of its discovery, to defy nature. There is, however, a price for this perceived invincibility.
“Stainless steel” is a broad term that encompasses many alloys. Fortunately, for the sake of this discussion, the “marine grades” consist of a fairly short list. Stainless steel is made up of primarily iron (much like ordinary mild steel). To this, varying amounts of chromium and nickel are added, depending on the grade being manufactured.
Nickel
Nickel in stainless steels promotes the stability of austenite, a marine grade of stainless steel. Austenite is stronger and more stable at higher temperatures than ferrite, yet another grade of stainless steel — one that is not well suited for marine use. Less nickel is needed to retain an austenitic structure as the nitrogen or carbon levels increase; when sufficient nickel is added to a chromium stainless steel, the structure changes from ferritic to austenitic.
Adding nickel improves toughness, ductility and weldability. Nickel increases resistance to oxidation, carburization, nitriding, thermal fatigue and strong acids, particularly reducing acids. It is a critically important alloying element in alloys used for corrosive and high-temperature applications.
These additions are what give stainless steel its unique corrosion-resistant (not corrosion-proof) properties. When exposed to the elements, specifically oxygen, the chromium enables this alloy to form a tough oxide film almost instantly. The result is what was once referred to as “rustless” or “rust-free” steel. But is this really accurate?
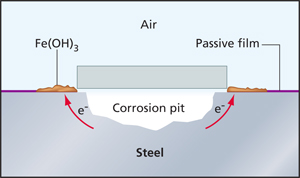 |
|
When stainless steel is exposed to air, a passive film forms that protects the metal. When air can’t get to an area, crevice corrosion can occur. |
As previously mentioned, there are several types of stainless steel, within which exist three subcategories: martensitic, which is often used for cooking utensils, rigging knives and other cutlery — essentially anything that requires hardening, like knives and tools; ferritic, which is used for auto trim and catalytic converters; and austenitic, which is used for marine applications, as well as buildings and structures (the Gateway Arch and art deco trim on NYC’s Chrysler Building, along with many kinds of flatware, are either made of or clad in austenitic stainless steel). Nickel is exclusive to the marine grades, which are exclusively in the “300 series” (more on that below). The alloying elements and the effect they have on the strength and corrosion resistance of stainless steel vary considerably within the grades of stainless. They include, in addition to chrome and nickel: sulfur, phosphorus, titanium, molybdenum, nitrogen and vanadium, as well as others. Thus, all stainless steels are not created equal.
The austenitic or marine grades of stainless steel are further distilled into the American Iron and Steel Institute’s (AISI) “300 series.” Where recreational and commercial vessels are concerned, this primarily means type 304 — sometimes referred to as 18-8, which reflects the percentage of alloying elements chrome and nickel, respectively — and type 316. The difference between these two is simply the amount of alloy added to the basic steel starting point: 304-series stainless steel consists of 18 to 20 percent chromium, and 8 to 12 percent nickel; 316-series stainless steel, on the other hand, consists of 16 to 18 percent chromium, and 8 to 12 percent nickel with an addition of 2 to 3 percent molybdenum. That final ingredient enables this grade of stainless steel to better resist a phenomenon referred to as crevice corrosion. Indeed, crevice corrosion is one of a few chinks in stainless steel’s armor, and among the most common failure mechanisms.
Much of the stainless steel that is readily available to the marine industry and boat owners is type 304, or 18-8. Technically, this is the “lowest” commonly available grade of marine-approved stainless steel. Its corrosion resistance is considerably higher than ordinary steel to be sure; however, contrary to popular belief, it’s far from corrosion-proof. Why then, the reader might ask, is it used, particularly if higher-quality, more corrosion-resistant options exist? As is often the case when utilizing a less-than-ideal product or material, it’s less expensive and it does a reasonably good job as long as it’s not called upon to do something for which it was not designed. As an aside, stainless steel, be it 304 or 316, that is to be welded must include the suffix “L” after its designating number, as in 304L or 316L. This denotes low carbon content, which helps to prevent weld migration, which will be discussed below.
There is yet another derivative of stainless steel that could be considered a super-corrosion-resistant metal of sorts. It’s commonly and often mistakenly referred to by its well-known trade name, Aquamet, which has become a generic name, much like Scotch Tape. Aquamet, however, is simply the most well known but not necessarily the most common form of this steel. It was one of the first proprietary stainless steel-based shafting alloys, though there are several others of equal quality.
With slight differences, these alloys possess the basic ingredients of 316 stainless steel, in addition to several other exotic elements such as tantalum, niobium and manganese. It’s used almost exclusively in the form of propeller and rudder shafts, although keel bolts and rigging (often referred to as “Nitronic,” another trade name) are also available in this or similar alloys, and it too comes in several grades (17, 19 and 22, the latter being the most resistant to corrosion and the only that is non-magnetic). In my experience, I have encountered countless severely corroded propeller shafts. I suspect, however, with some exceptions, the ones that do corrode are made of lower-grade shaft alloys, specifically 17, 19 or less exotic (and thus less corrosion-resistant) non-proprietary shaft alloys.
There’s a price to be paid for supreme resistance to corrosion, literally. As one might expect, super-corrosion-resistant alloys come with a higher although typically justifiable price tag. Nevertheless, they still require cathodic protection in the form of zinc, magnesium (for fresh water only) or aluminum anodes.
Problems with stainless
Bearing in mind all of the advantages of stainless steel, it might seem — and the prevailing notion among boaters would support this — that stainless steel is virtually indestructible. Regrettably, it’s not. While there are several types of common corrosion, stainless steel in the marine world suffers primarily from two types: crevice corrosion and carbide precipitation. The most common, or the highest profile among these, is crevice corrosion.
Crevice or pitting corrosion occurs when stainless steel is exposed to an oxygen-poor environment for an extended period. The tough, nearly impenetrable oxide film that forms as soon as stainless steel is exposed to air (the term “inox,” seen on many European stainless components, is simply an abbreviation for inoxidizable, which refers to stainless steel’s resistance to staining) only remains intact as long as the metal is exposed to oxygen (or dissolved oxygen found in moving water). If stainless steel is placed in an environment where it is starved of oxygen and it is exposed to water, either fresh or salt, it becomes susceptible to crevice corrosion, which typically manifests itself as roughness, valleys, pitting or even worm-like holes (it’s the chlorides in seawater that accelerate the destruction of the oxide layer). It’s readily replaced as long as oxygen is present. However, when oxygen is not present, the chlorides win this tug of war, the oxide layer diminishes and, from a corrosion point of view, stainless steel begins to behave more like mild steel. Prop shaft manufacturers test their alloy’s crevice corrosion resistance by placing a rubber band around a representative section and then submersing it in hot salt water for an extended period.
 |
 |
|
 |
 |
|
|
Examples of stainless corrosion are shown here, including corrosion under fasteners and backing plates, corrosion on a prop shaft and a corroded link on a steering system chain. |
||
The most likely location for crevice corrosion to occur is where stainless alloys are used for raw-water and underwater hardware, which may include propeller shafts, struts, raw-water intake, discharge and sanitation plumbing, exhaust systems (particularly diesel exhaust because of the sulfur content of diesel fuel, which becomes acidic when mixed with water), shaft logs and fasteners, or nuts, bolts and screws that are used below the waterline to secure hardware such as struts, seacocks, hull strainers, swim platform supports, etc.
Because nearly all commonly available stainless-steel fasteners are made of 304 alloy, they run an exceptionally high risk of suffering from crevice corrosion when used in applications below the waterline or in splash zones (they are particularly prone to corrosion where they cannot be easily inspected, where they pass through the hull for instance). And the exposure to water does not necessarily require immersion: Stainless steel that is imbedded in hygroscopic materials such as wood, foam, balsa core or even fiberglass may suffer from crevice corrosion.
Stagnant or still water
It is preferable to avoid using stainless steel in applications where it is regularly exposed to stagnant or still water for extended periods. Ideally, high-quality silicon bronze, cupronickel, Inconel 625, Monel or non-metallic alternatives should be used, particularly for fasteners and raw-water plumbing. The fasteners used to secure struts, strainers and other underwater hardware, for instance, should be bronze (true zinc-free bronze, not brass) rather than stainless steel. If stainless steel must be used in such an application, for a custom-made strut or shaft log for instance, it should be fabricated from the most corrosion-resistant, readily weldable marine alloy available — 316L or 317L — primed, antifouled and provided with cathodic protection.
Propeller shafts fall into a special category. Because they require both exceptional strength and corrosion resistance, propeller shafts are typically fabricated from a proprietary stainless alloy that includes a host of other trace elements. These mixtures yield the ideal combination for the environment in which shafts operate. The aforementioned trade names such as Aquamet, Aquatec and Aqualoy, among others, are available in several strength and corrosion resistance grades. The most corrosion-resistant of these, and therefore desirable particularly for recreational applications where use and oxygen exposure is limited, is designated as 22. Shafts made of this alloy and grade are as close as a boatbuilder can get to corrosion-proof where steel-based alloys are concerned. When purchasing such a shaft, ask the vendor to verify the alloy grade and manufacturer. Shaft alloy grades 17 and 19 are magnetic, while 22 is non-magnetic.
Shafts manufactured from ordinary, non-shaft alloy stainless steel, on the other hand, frequently suffer from crevice corrosion attacks, often beneath shaft bearings and stuffing box packing — again, where it cannot be easily observed.
In order to avoid corrosion and failures, only use stainless steel when and where it’s best suited for the job. For submerged applications, bronze is preferred; however, if stainless steel must be used, be sure to use nothing less than 316-series alloys.
Steve D’Antonio is a marine writer and photographer, ABYC Master Technician and owns Steve D’Antonio Marine Consulting (stevedmarineconsulting.com).

